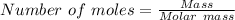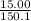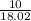Chemistry, 24.03.2020 05:29 lberman2005p77lfi
15.00 g of aluminum sulfide (150.1 g/mol) and 10.00 g of water (18.02 g/mol) react until the limiting reactant is used up. Calculate the mass of H2S (34.08 g/mol) that can be produced from these reactants. Notice that you will need to balance the reaction equation.
___Al2S3(s)+ ___H2O > ___Al(OH)3(s)+ ___H2S(g)
a. 13.89 g
b. 10.21 g
c. 19.67 gd. 9.456 g
e. 1.108 g

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, Ashleyvasquez2261
Type the letter that represents the correct location for each particle type below.
Answers: 1


Chemistry, 22.06.2019 19:20, johnkings140
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 20:50, iluminatioffial9699
One nanometer is equal to how many meters?
Answers: 2
You know the right answer?
15.00 g of aluminum sulfide (150.1 g/mol) and 10.00 g of water (18.02 g/mol) react until the limitin...
Questions in other subjects:







Mathematics, 09.11.2020 17:20


Chemistry, 09.11.2020 17:20