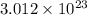Chemistry, 24.03.2020 01:27 juanesmania
When an ionic compound such as sodium chloride (NaCl) is placed in water the component atoms of the NaCl crystal dissociate into individual sodium ions (Na+) and chloride ions (Cl-). In contrast, the atoms of covalently bonded molecules (e. g., glucose, sucrose, glycerol) do not generally dissociate when placed in aqueous solution. Which of the following solutions would be expected to contain the greatest number of particles (molecules or ions)? A) 1 L of 0.5 M NaCl B) 1 L of 0.5 M glucose C) 1 L of 1.0 M NaCl D) 1 L of 1.0 M glucose E) C and D will contain equal numbers of particles.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, azzyla2003
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 22.06.2019 17:00, davisnaziyahovz5sk
The arrangement of particles is most ordered in a sample of
Answers: 1

Chemistry, 22.06.2019 20:00, AaronEarlMerringer
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
You know the right answer?
When an ionic compound such as sodium chloride (NaCl) is placed in water the component atoms of the...
Questions in other subjects:


Mathematics, 06.05.2021 19:40

Chemistry, 06.05.2021 19:40


Chemistry, 06.05.2021 19:40

English, 06.05.2021 19:40

Mathematics, 06.05.2021 19:40

History, 06.05.2021 19:40


 number of molecules/ions
number of molecules/ions number of ions
number of ions volume) no. of particles
volume) no. of particles