
Chemistry, 24.03.2020 00:56 arielmajesty9107
The two reactions involved in quantitatively determining the amount of iodate in solution are: IO3-(aq) + 5 I-(aq) + 6 H+(aq) --> 3 I2(aq) + 3 H2O(l) followed by reaction of the I2: I2(aq) + 2 S2O32- --> 2 I-(aq) + S4O62-(aq). What is the stoichiometric factor, that is the number of moles of Na2S2O3 reacting with one mole of KIO3?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, emilyswinge4421
Listenbase your answer to the question on the information below. nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body. cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment. which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2


Chemistry, 23.06.2019 09:00, aaronroberson4940
Weight is a measure of: inertia force matter mass
Answers: 1
You know the right answer?
The two reactions involved in quantitatively determining the amount of iodate in solution are: IO3-(...
Questions in other subjects:


Mathematics, 07.10.2021 01:00


Mathematics, 07.10.2021 01:00

Mathematics, 07.10.2021 01:00


Medicine, 07.10.2021 01:00


Engineering, 07.10.2021 01:00

Mathematics, 07.10.2021 01:00

 reacting with one mole of
reacting with one mole of  .
.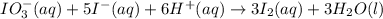 ..[1]
..[1] ..[2]
..[2]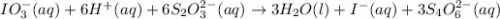
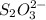 reacts with 1 mole of
reacts with 1 mole of 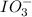 .
.

