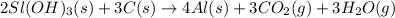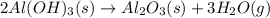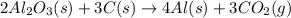Chemistry, 21.03.2020 10:56 jakhunter354
The extraction of aluminum metal from the aluminum hydroxide in bauxite ore by the Hall-Héroult process is one of the most remarkable success stories of late 19th century practical chemistry, turning aluminum from a rare and precious metal into the cheap commodity it is today. In the first step, aluminum hydroxide reacts to form alumina and water: 2AlOH3(s)→Al2O3(s)+3H2O(g) In the second step, alumina and carbon react to form aluminum and carbon dioxide: 2Al2O3(s)+3C(s)→4Al(s)+3CO2(g) Write the net chemical equation for the production of aluminum from aluminum hydroxide and carbon. Be sure your equation is balanced.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, asianaenaeh
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 12:00, shifaxoxoxo
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2

Chemistry, 23.06.2019 04:10, nabeelunique
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2
You know the right answer?
The extraction of aluminum metal from the aluminum hydroxide in bauxite ore by the Hall-Héroult proc...
Questions in other subjects:

Biology, 08.10.2019 15:20

Mathematics, 08.10.2019 15:20





Spanish, 08.10.2019 15:20


Physics, 08.10.2019 15:20