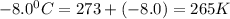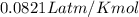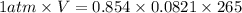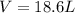Chemistry, 20.03.2020 10:04 brandon56238
A reaction at −8.0°C evolves 854.mmol of boron trifluoride gas. Calculate the volume of boron trifluoride gas that is collected. You can assume the pressure in the room is exactly 1atm. Be sure your answer has the correct number of significant digits.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, FloweyFlower
Aballoon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon? a balloon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon?
Answers: 3

Chemistry, 22.06.2019 03:00, actheorian8142
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 05:00, adrian128383
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 20:00, teacherpreacher
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
A reaction at −8.0°C evolves 854.mmol of boron trifluoride gas. Calculate the volume of boron triflu...
Questions in other subjects:


Engineering, 19.03.2021 23:20


Physics, 19.03.2021 23:20



History, 19.03.2021 23:20

Biology, 19.03.2021 23:20

Mathematics, 19.03.2021 23:20