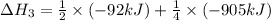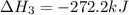Chemistry, 20.03.2020 07:19 valeriegarcia12
Nitric oxide (NO) can be formed from nitrogen, hydrogen and oxygen in two steps. In the first step, nitrogen and hydrogen react to form ammonia:
N2(g) + 3H2(g) →2NH3(g)
ΔH=−92.kJ
In the second step, ammonia and oxygen react to form nitric oxide and water:
4NH3(g) + 5O2(g) → 4NO(g) +6H2O(g)
ΔH=−905.kJ
Calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions.

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 11:00, hannah5143
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

Chemistry, 22.06.2019 11:20, Jessicadiaz8602
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1
You know the right answer?
Nitric oxide (NO) can be formed from nitrogen, hydrogen and oxygen in two steps. In the first step,...
Questions in other subjects:



Mathematics, 29.07.2020 18:01





English, 29.07.2020 18:01


Mathematics, 29.07.2020 18:01

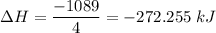
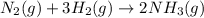 (1)
(1)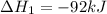
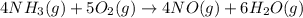 (2)
(2)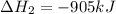
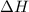 for the following reaction i.e,
for the following reaction i.e,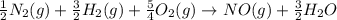
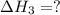
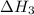 for the reaction will be:
for the reaction will be: