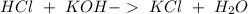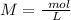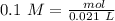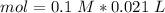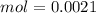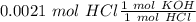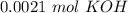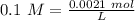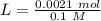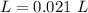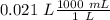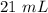Chemistry, 20.03.2020 03:34 emileehogan
Two 21.0 mL samples, one 0.100 M HCl and the other 0.100 M HF, were titrated with 0.200 M KOH. Answer each of the following questions regarding these two titrations. You may want to reference (Pages 755 - 769) Section 17.4 while completing this problem. Part A What is the volume of added base at the equivalence point for HCl

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, ayoismeisalex
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2



Chemistry, 22.06.2019 08:30, ayaanwaseem
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2
You know the right answer?
Two 21.0 mL samples, one 0.100 M HCl and the other 0.100 M HF, were titrated with 0.200 M KOH. Answe...
Questions in other subjects:






Social Studies, 21.08.2019 11:00


Biology, 21.08.2019 11:00


Mathematics, 21.08.2019 11:00