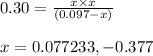For the reaction POCl3(g) ⇀↽ POCl(g) + Cl2(g) Kc = 0.30. An initial 0.3 moles of POCl3 are placed in a 3.1 L container with initial concentrations of POCl and Cl2 equal to zero. What is the final concentration of POCl3? 1. final concentration = 0.281774 M 2. final concentration = 0.077 M 3. final concentration = 0.185 M 4. final concentration = 0.039534 M 5. final concentration = 0.019767 M

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

You know the right answer?
For the reaction POCl3(g) ⇀↽ POCl(g) + Cl2(g) Kc = 0.30. An initial 0.3 moles of POCl3 are placed in...
Questions in other subjects:

Mathematics, 08.03.2020 04:02



Mathematics, 08.03.2020 04:03





Mathematics, 08.03.2020 04:04

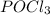 is 0.019767 M
is 0.019767 M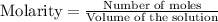


 for above equation follows:
for above equation follows:![K_c=\frac{[POCl][Cl_2]}{[POCl_3]}](/tpl/images/0555/1089/0f7c6.png)