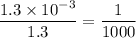Chemistry, 20.03.2020 01:06 blondielocks2002
Explain why a solution that is 1.3 M HF and 1.3 mM KF is not a good buffer. HF is a strong acid and cannot be used in a buffer system. The ratio of acid to conjugate base is outside the buffer range of 10:1. The two species are not a conjugate acid base pair. KF is not soluble in water and cannot be used in a buffer system.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, ethanw8973
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 16:00, winnie45
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2
You know the right answer?
Explain why a solution that is 1.3 M HF and 1.3 mM KF is not a good buffer. HF is a strong acid and...
Questions in other subjects:

History, 15.01.2020 19:31



English, 15.01.2020 19:31

Business, 15.01.2020 19:31

Health, 15.01.2020 19:31




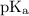 + log
+ log 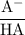
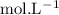 in HF and 1.3
in HF and 1.3 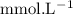 in KF, the ratio is:
in KF, the ratio is: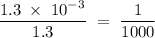
![\text{pH} = \text{pK}_{\text{a}} + \log\dfrac{\text{[A$^{-}$]}}{\text{[HA]}}](/tpl/images/0555/0502/0e377.png)
![\dfrac{1}{10} \leq \dfrac{\text{[A$^{-}]$}}{\text{[HA]}} \leq \dfrac{10}{1}](/tpl/images/0555/0502/63cff.png)