
Chemistry, 20.03.2020 00:06 buddyclayjohnson
A mixture of methane (CH4) and ethane (C2H6) is stored in a container at 294 mm Hg. The gases are burned in air to form CO2 and H2O. If the pressure of CO2 is 351 mm Hg measured at the same temperature and volume as the original mixture, calculate the mole fraction of the gases.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:30, Kiaraboyd9366
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1

Chemistry, 22.06.2019 21:30, sullivanjakob
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1

Chemistry, 23.06.2019 00:00, kittenalexis68
How many atoms or molecules are there in a mole of a substance?
Answers: 1

Chemistry, 23.06.2019 01:30, Nathaliasmiles
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2
You know the right answer?
A mixture of methane (CH4) and ethane (C2H6) is stored in a container at 294 mm Hg. The gases are bu...
Questions in other subjects:

Mathematics, 16.02.2021 01:00







History, 16.02.2021 01:00


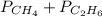 = 294 mm Hg
= 294 mm Hg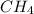 will yield 1 mole of
will yield 1 mole of 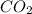 whereas 1 mole of
whereas 1 mole of 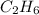 will yield 2 moles of
will yield 2 moles of 
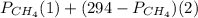 = 351 mm Hg
= 351 mm Hg = 2(294) - 351
= 2(294) - 351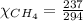
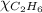 = 1 - 0.806
= 1 - 0.806


