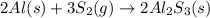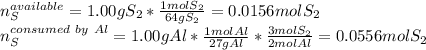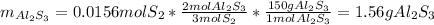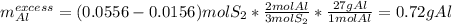Chemistry, 19.03.2020 21:45 2023jpeterson
Aluminum reacts with sulfur gas to produce aluminum sulfide. a) What is the limiting reactant? What is the excess reagent? b) How many grams of Aluminum Sulfide will be produced? c) How many grams of the excess reactant will be left over in the reaction?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:20, rscvsdfsrysas1857
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 22.06.2019 18:20, juansebas35
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2
You know the right answer?
Aluminum reacts with sulfur gas to produce aluminum sulfide. a) What is the limiting reactant? What...
Questions in other subjects:







Mathematics, 07.10.2019 20:00