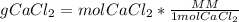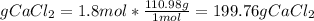Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:30, junkmailemail42
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

You know the right answer?
In a group assignment, students are required to fill 10 beakers with 0.720 M CaCl2. If the molar mas...
Questions in other subjects:



Mathematics, 16.05.2021 02:50


History, 16.05.2021 02:50

Biology, 16.05.2021 02:50


Mathematics, 16.05.2021 02:50

Physics, 16.05.2021 02:50

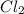 and dilute it in a flask of 2.5 L.
and dilute it in a flask of 2.5 L.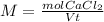
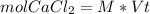
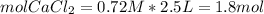
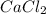 in grams of
in grams of