
Chemistry, 19.03.2020 10:33 maggie2018
4Fe +302–> 2Fe2O3 how many moles of Fe2O3 are produced when 0.27 moles of Fe is reacted?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 03:00, BeeShyanne
Use the half-reactions of the reaction au(oh)3 + hi -> au +i2 +h2o to answer the questions
Answers: 1

Chemistry, 23.06.2019 04:50, mia36492
The diagin dilutepage 6 of 12a6a5(a)fluorine, chlorine, bromine and iodine are placed in the same group of theperiodic table. state the common name used to describe elements in this group.(i)state the group in which the elements are placed and explain whythey are placed in that group.(ii)which of the above named elements is a solid at roomtemperature and pressure?
Answers: 2

Chemistry, 23.06.2019 10:10, nancysue1975
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3
You know the right answer?
4Fe +302–> 2Fe2O3 how many moles of Fe2O3 are produced when 0.27 moles of Fe is reacted?...
Questions in other subjects:



Mathematics, 20.10.2020 01:01


Mathematics, 20.10.2020 01:01

English, 20.10.2020 01:01



Biology, 20.10.2020 01:01

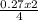 = 0.14 moles of Fe₂O₃
= 0.14 moles of Fe₂O₃


