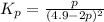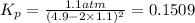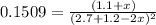Chemistry, 19.03.2020 09:36 TanishaSchollaert1
Nitrogen dioxide is one of the many oxides of nitrogen (often form another form of NOx, dinitrogen tetroxide A chemical engineer studying this reaction fils a 500. mL flask at 7.9 °C with 4.9 atm of nitrogen dioxide gas. He thèn raises the temperature considerably and when the mixture has come to equilibrium determines that it contains 2.7 atm of nitrogen dioxide gas The engineer then adds another 1.2 atm of nitrogen dioxide, and allows the mixture to come to equilibrium again. Calculate the pressure of dinitrogen tetroxide after equilibrium is reached the second time. Round your answer to 2 significant digits collectively called·N ' that are of interest to atmospheric chemistry. It can eact with ter to 囲 atm □-10 I Don't Know Submit

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:30, megaaan214p61pb7
Which compounds have the empirical formula ch2o? a. c2h4o2 b. c3h6o3 c. ch2o2 d. c5h10o5 e. c6h12o6
Answers: 3


Chemistry, 22.06.2019 22:00, aliciaa101
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1
You know the right answer?
Nitrogen dioxide is one of the many oxides of nitrogen (often form another form of NOx, dinitrogen t...
Questions in other subjects:


Chemistry, 01.07.2019 00:50

Computers and Technology, 01.07.2019 00:50

Mathematics, 01.07.2019 00:50

Biology, 01.07.2019 00:50

Mathematics, 01.07.2019 00:50

Social Studies, 01.07.2019 00:50




 :
: