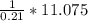A gas contains 75.0 wt% methane, 10.0% ethane, 5.0% ethylene, and the balance water. (a) Calculate the molar composition of this gas on both awet and a dry basis and the ratio (mol H2O/ mol dry gas). (b) If100kg/30%excessair,(kmol/ h)? How would the answer change if the combustion were only 75% complete?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, 2019reynolds
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1


Chemistry, 22.06.2019 11:00, RidhaH
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural. question 2 reflects a moral or social value. question 3 refers to something that can be measured. question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 23.06.2019 06:20, raidattarab
What is the magnitude of the force of gravity between to 1000 kg cars which are separated by distance of 25. 0 km on an interstate highway? the force between the two cars will be what
Answers: 3
You know the right answer?
A gas contains 75.0 wt% methane, 10.0% ethane, 5.0% ethylene, and the balance water. (a) Calculate t...
Questions in other subjects:


Mathematics, 29.11.2020 20:50



Mathematics, 29.11.2020 20:50



Mathematics, 29.11.2020 20:50

Mathematics, 29.11.2020 20:50

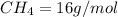
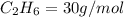
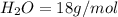
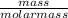
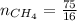
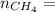 4.69 moles
4.69 moles 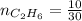
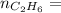 0.33 moles
0.33 moles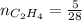
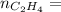 0.18 moles
0.18 moles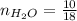
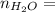 0.56 moles
0.56 moles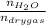
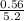




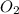 required = (2 × 4.69) + (3.5 × 0.33) + (3 × 0.18) k moles
required = (2 × 4.69) + (3.5 × 0.33) + (3 × 0.18) k moles