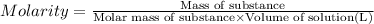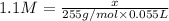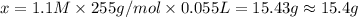Chemistry, 19.03.2020 05:02 22moneymorgan
A chemist adds of a barium acetate solution to a flask. Calculate the mass in grams of barium acetate the chemist has added to the flask. Be sure your answer has the correct number of significant digits.

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 06:00, rebeccacruzz2017
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
A chemist adds of a barium acetate solution to a flask. Calculate the mass in grams of barium acetat...
Questions in other subjects:




Mathematics, 16.10.2019 00:00

Social Studies, 16.10.2019 00:00

History, 16.10.2019 00:00


Mathematics, 16.10.2019 00:00

Mathematics, 16.10.2019 00:00