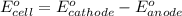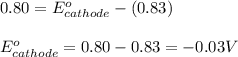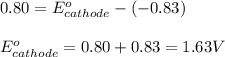Chemistry, 19.03.2020 03:59 hardwick744
A certain half-reaction has a standard reduction potential E⁰ʀᴇᴅ = 0.83 V. An engineer proposes using this half-reaction at the anode of a galvanic cell that must provide at least 0.80V of electrical power. The cell will operate under standard conditions. Note for advanced students: assume the engineer requires this half-reaction to happen at the anode of the cell.
(1) Is there a minimum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, write "yes" and calculate the minimum. Round your answer to 2 decimal places. If there is no lower limit, write "no".
(2) Is there a maximum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, write "yes" and calculate the minimum. Round your answer to 2 decimal places. If there is no lower limit, write "no".

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, jwood287375
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3


Chemistry, 22.06.2019 23:00, lilsnsbsbs
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2

Chemistry, 23.06.2019 00:10, Rubendelarosa1529
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3
You know the right answer?
A certain half-reaction has a standard reduction potential E⁰ʀᴇᴅ = 0.83 V. An engineer proposes usin...
Questions in other subjects:

Mathematics, 20.05.2020 21:57


Mathematics, 20.05.2020 21:57

Mathematics, 20.05.2020 21:57


English, 20.05.2020 21:57

Health, 20.05.2020 21:57

Mathematics, 20.05.2020 21:57


Spanish, 20.05.2020 21:57

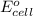 of the reaction, we use the equation:
of the reaction, we use the equation: