
Chemistry, 19.03.2020 01:08 ayoismeisjjjjuan
The Haber reaction for the manufacture of ammonia is: N2 + 3H2 → 2NH3 Without doing any experiments, which of the following can you say MUST be true? Disappearance rate of H2 = 3 (Disappearance rate of N2). The reaction is first order in N2. Reaction rate = -Δ[N2]/Δt. The reaction is not an elementary reaction. Δ[H2]/Δt will have a positive value. Disappearance rate of N2 = 3 (Disappearance rate of H2). The activation energy is positive.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:40, Islandgirl67
If equal masses of the listed metals were collected , which would have a greatest volume ? a. aluminum 2.70,b. zinc7.14,c. copper 8.92,d. lead 11.34
Answers: 2

Chemistry, 22.06.2019 03:00, ian2006huang
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 16:40, roderickhinton
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3
You know the right answer?
The Haber reaction for the manufacture of ammonia is: N2 + 3H2 → 2NH3 Without doing any experiments,...
Questions in other subjects:

English, 12.02.2021 06:30






Mathematics, 12.02.2021 06:30


History, 12.02.2021 06:30


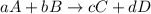
![\text{Rate of disappearance of A}=-\frac{1}{a}\frac{d[A]}{dt}](/tpl/images/0553/1420/2d8eb.png)
![\text{Rate of disappearance of B}=-\frac{1}{b}\frac{d[B]}{dt}](/tpl/images/0553/1420/1e77e.png)
![\text{Rate of formation of C}=+\frac{1}{c}\frac{d[C]}{dt}](/tpl/images/0553/1420/cee4b.png)
![\text{Rate of formation of D}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0553/1420/7ef32.png)
![Rate=-\frac{1}{a}\frac{d[A]}{dt}=-\frac{1}{b}\frac{d[B]}{dt}=+\frac{1}{c}\frac{d[C]}{dt}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0553/1420/d4b94.png)
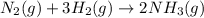
![\text{Rate of disappearance of }N_2=-\frac{d[N_2]}{dt}](/tpl/images/0553/1420/25b13.png)
![\text{Rate of disappearance of }H_2=-\frac{1}{3}\frac{d[H_2]}{dt}](/tpl/images/0553/1420/ebff2.png)
![\text{Rate of formation of }NH_3=+\frac{1}{2}\frac{d[NH_3]}{dt}](/tpl/images/0553/1420/f55ec.png)



