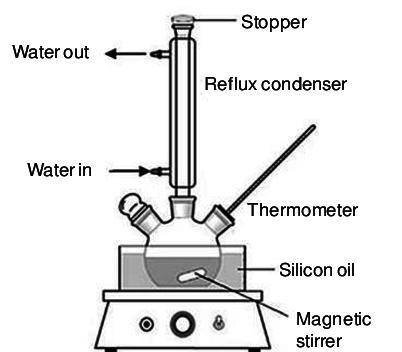
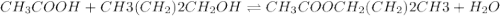Which one of the following choices describes most accurately the actual, internal reaction temperature (in other words, the temperature of the reaction mixture inside the reaction vial) for the Fischer esterification experiment of 1-butanol with acetic acid to form n-butyl acetate? Select one, and explain your answer.
a) Sand bath temperature (160-180 °C)
b) Boiling point of 1-butanol (116-118 °C)
c) Boiling point of the reaction mixture (reflux temperature)
d) Boiling point of acetic acid (117 °C)
e) Boiling point of n-butyl acetate (124-126 °C)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, MrSavannahCat
Which produce would best increase the amount of heat energy that is actually gained by calorimeter b
Answers: 1


Chemistry, 22.06.2019 10:30, kluckey3426
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 22:30, robertss403
How many moles of kci are produced from 2.50 moles k
Answers: 1
You know the right answer?
Which one of the following choices describes most accurately the actual, internal reaction temperatu...
Questions in other subjects:

Mathematics, 04.02.2020 11:51


Biology, 04.02.2020 11:51


Biology, 04.02.2020 11:51


Mathematics, 04.02.2020 11:51

History, 04.02.2020 11:51

Physics, 04.02.2020 11:51