Chemistry, 18.03.2020 21:58 emily965692
Manganese commonly occurs in nature as a mineral. The extraction of manganese from the carbonite mineral rhodochrosite, involves a two-step process. In the first step, manganese (II) carbonate and oxygen react to form manganese (IV) oxide and carbon dioxide: 2MnCO3(s)+O2(g)→2MnO2(s)+2CO2(g) In the second step, manganese (IV) oxide and aluminum react to form manganese and aluminum oxide: 3MnO2(s)+4Al(s)→3Mn(s)+2Al2O3(s) Write the net chemical equation for the production of manganese from manganese (II) carbonate, oxygen and aluminum. Be sure your equation is balanced.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, BrandyLeach01
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3

Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 01:00, jaidencoolman2510
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 04:00, clickbaitdxl
What two categories of toxins were present in the air at dish, texas as a result of the gas pipelines that pass through the area
Answers: 1
You know the right answer?
Manganese commonly occurs in nature as a mineral. The extraction of manganese from the carbonite min...
Questions in other subjects:

Biology, 07.10.2019 16:30



History, 07.10.2019 16:30






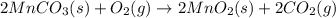 ( × 3)
( × 3)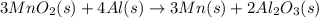 ( × 2)
( × 2)