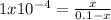Chemistry, 18.03.2020 19:39 JellalFernandes
A reaction A(g)⇌B(g)has an equilibrium constant of 1.0×10−4 For which of the initial reaction mixtures is the small approximation most likely to apply? Explain why.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, thatonestudent2271
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 05:30, medlinalex
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 21:20, 50057543
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

Chemistry, 23.06.2019 00:30, evelynalper08
Which radioisotope is used to date fossils? a. oxygen-16 b. carbon-14 c. uranium-238 d. carbon-12
Answers: 2
You know the right answer?
A reaction A(g)⇌B(g)has an equilibrium constant of 1.0×10−4 For which of the initial reaction mixtur...
Questions in other subjects:







Social Studies, 03.12.2020 07:10

Mathematics, 03.12.2020 07:10

Mathematics, 03.12.2020 07:10

Mathematics, 03.12.2020 07:10

 in this case would be 0.00001M which is the smallest one from the given options, considering:
in this case would be 0.00001M which is the smallest one from the given options, considering: