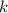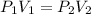A certain amount of chlorine gas was placed inside a cylinder with a movable piston at one end. The initial volume was 3.00 L and the initial pressure of chlorine was 1.85 atm . The piston was pushed down to change the volume to 1.00 L. Calculate the final pressure of the gas if the temperature and number of moles of chlorine remain constant

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:30, damienlopezram
You 4. you have been swimming in your neighbor’s pool for an hour. the relative humidity of the air is 30 percent. will you feel warm or cool when you step out of the pool? explain your answer.
Answers: 1

Chemistry, 21.06.2019 23:00, brapmaster764
What is the formula that this ionic compounds could form sr2+p3-o2-
Answers: 3

Chemistry, 22.06.2019 07:20, mathman783
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 10:00, aschool2000
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2
You know the right answer?
A certain amount of chlorine gas was placed inside a cylinder with a movable piston at one end. The...
Questions in other subjects:

Mathematics, 29.11.2021 21:10

Mathematics, 29.11.2021 21:10


Chemistry, 29.11.2021 21:10

Mathematics, 29.11.2021 21:10

Mathematics, 29.11.2021 21:10




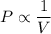
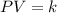 Pressure multiplied by volume equals some constant
Pressure multiplied by volume equals some constant