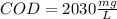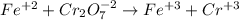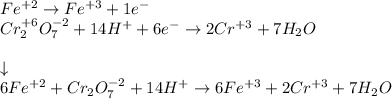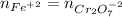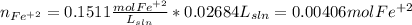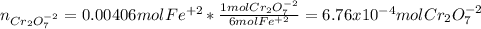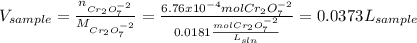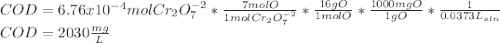Your standard iron solution is 0.1511 M Fe(II), your dichromate solution is 0.0181 M dichromate, and it took 26.84 mL of your standard iron solution to titrate the excess dichromate in your unknown. What is the chemical oxygen demand of the sample, in units of mg O2/L? (Report your answer with 4 sig figs)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, brasherfamily14
Which term describes a fracture in the earth at which land stays in the same place? a. joint b. fault c. split d. hinge
Answers: 1


Chemistry, 22.06.2019 09:00, triddi666
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 23.06.2019 03:30, alvfran1041
Astudent uses universal ph paper to find the ph of three solutions . solution a has a ph of 5 solution b has a ph of 11 and solution c has a ph of 7 identify which solution is acidic which solution is neutral and which solution is basic
Answers: 1
You know the right answer?
Your standard iron solution is 0.1511 M Fe(II), your dichromate solution is 0.0181 M dichromate, and...
Questions in other subjects:

History, 11.06.2020 02:57

Business, 11.06.2020 02:57


Mathematics, 11.06.2020 02:57

English, 11.06.2020 02:57


Biology, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57