
Chemistry, 17.03.2020 00:27 aliyyahlove
In the Haber process, ammonia is synthesized from nitrogen and hydrogen: N2(g) + 3H2(g) → 2NH3(g) ΔG° at 298 K for this reaction is -33.3 kJ/mol. The value of ΔG at 298 K for a reaction mixture that consists of 1.9 atm N2, 1.6 atm H2, and 0.65 atm NH3 is .

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:30, kevinh2683
Apump contains 0.5 l of air at 203 kpa. you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2



Chemistry, 23.06.2019 03:30, cupcake3103670
Name 3 types of energy you see being used as you look around a classroom
Answers: 1
You know the right answer?
In the Haber process, ammonia is synthesized from nitrogen and hydrogen: N2(g) + 3H2(g) → 2NH3(g) ΔG...
Questions in other subjects:

Mathematics, 04.03.2021 21:20


Mathematics, 04.03.2021 21:20

Mathematics, 04.03.2021 21:20


Mathematics, 04.03.2021 21:20




Mathematics, 04.03.2021 21:20

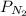 = 1.9 atm
= 1.9 atm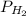 = 1.6 atm
= 1.6 atm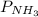 = 0.65 atm
= 0.65 atm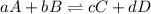
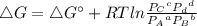
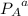 = Partial presser of A
= Partial presser of A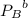 =Partial presser of B
=Partial presser of B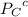 =Partial presser of C
=Partial presser of C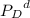 = Partial presser of D
= Partial presser of D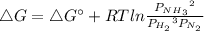
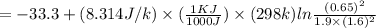 -
-

