
Chemistry, 16.03.2020 23:45 alejandramirand9836
A cylinder with a moveable piston contains 92g of Nitrogen. The external pressure is constant at 1.00 atm. The initial temperature is 200K. When the temperature is decreased by 85 K, by putting it in a lower temperature freezer, the volume will decrease, according to the Ideal Gas Law. Calculate the work for this process. Express your answer in J. The conversion factor between liter atmospheres and joules is 101.3 J

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, megaaan214p61pb7
Which compounds have the empirical formula ch2o? a. c2h4o2 b. c3h6o3 c. ch2o2 d. c5h10o5 e. c6h12o6
Answers: 3

Chemistry, 22.06.2019 12:30, johnsont8377
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1

You know the right answer?
A cylinder with a moveable piston contains 92g of Nitrogen. The external pressure is constant at 1.0...
Questions in other subjects:


English, 11.12.2020 04:00



Mathematics, 11.12.2020 04:00




Mathematics, 11.12.2020 04:00

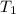 = 200 K
= 200 K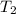 = 200 - 85 = 115 K
= 200 - 85 = 115 K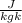
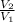 =
= 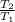 ------------ ( 1 )
------------ ( 1 )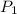
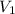 = m R
= m R 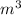
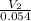 =
= 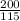
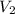 = 0.094
= 0.094 

