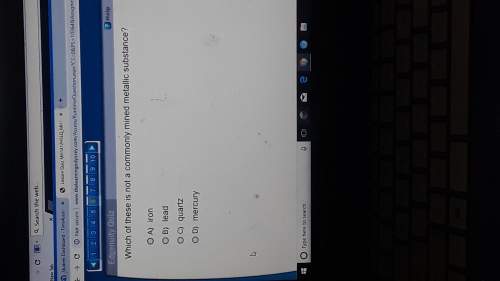
Chemistry, 16.03.2020 23:00 deannajd03
Write the dissolution reaction for manganese(II) chloride in water. Use the pull-down menus to specify the state of each reactant and product. + Is manganese(II) chloride considered soluble or not soluble ? ... ... A. Soluble ... B. Not soluble Based upon this, the equilibrium constant for this reaction will be: ... ... A. Greater than 1 ... B. Less than 1

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, Lesquirrel
Acar tire has a pressure of 2.38 atm at 15.2 c. if the pressure inside reached 4.08 atm, the tire will explode. how hot would the tire have to get for this to happen? report the temperature in degrees celsius.
Answers: 2


You know the right answer?
Write the dissolution reaction for manganese(II) chloride in water. Use the pull-down menus to speci...
Questions in other subjects:


Chemistry, 14.04.2020 19:32