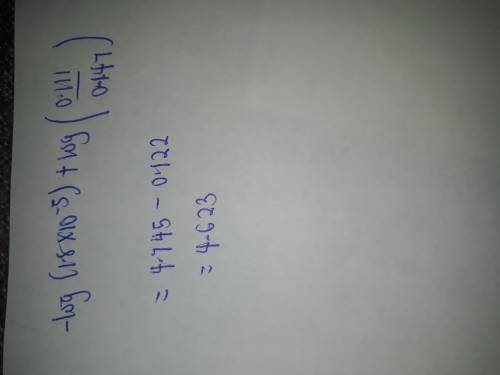Chemistry, 16.03.2020 21:31 ayoismeisalex
Calculate the pH after 0.018 mole of HCl is added to 1.00 L of each of the four solutions. (Assume that all solutions are at 25°C.) (a) 0.129 M acetic acid (HC2H3O2, Ka = 1.8 ✕ 10−5) (b) 0.129 M sodium acetate (NaC2H3O2) (c) pure H2O (d) 0.129 M HC2H3O2 and 0.129 M NaC2H3O2

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, scottbrandon653
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 05:40, timmonskids6027
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

Chemistry, 22.06.2019 09:10, GreatBaconGamer
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1

Chemistry, 22.06.2019 21:00, rah45
Which of these is an example of pseudoscience? a) predicting the time of sunrise based on data on position of earth b) predicting the date of the moon phases based on data on position of earth c) predicting eclipses based on the position of the sun and the moon d) predicting future events in a person's life based on the position of the moon
Answers: 1
You know the right answer?
Calculate the pH after 0.018 mole of HCl is added to 1.00 L of each of the four solutions. (Assume t...
Questions in other subjects:

History, 19.01.2021 14:00


Mathematics, 19.01.2021 14:00



Mathematics, 19.01.2021 14:10

English, 19.01.2021 14:10

English, 19.01.2021 14:10