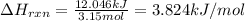3.15 mol of an unknown solid is placed into enough water to make 150.0 mL of solution. The solution's temperature increases by 16.01°C. Calculate ∆H for the dissolution of the unknown solid. (The specific heat of the solution is 4.18 J/g・°C and the density of the solution is 1.20 g/mL).

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2


Chemistry, 23.06.2019 01:00, Angelofpink1143
If i had 2 m naoh solution, what does the 2 m stand for? 2 molar, but 2 of a solute in 1
Answers: 1
You know the right answer?
3.15 mol of an unknown solid is placed into enough water to make 150.0 mL of solution. The solution'...
Questions in other subjects:


Mathematics, 13.11.2021 05:20



SAT, 13.11.2021 05:20

Mathematics, 13.11.2021 05:20



English, 13.11.2021 05:20

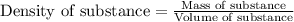
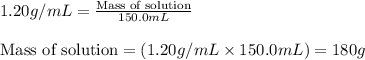
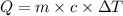
 = change in temperature = 16.01°C
= change in temperature = 16.01°C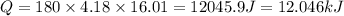
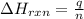
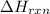 = enthalpy change of the reaction
= enthalpy change of the reaction