Show Your Work
2SO3(g) -> 2SO2(g)+O2(g)
Calculate Keq for this reaction if the eq...

Chemistry, 16.03.2020 20:43 lelseymota123
Show Your Work
2SO3(g) -> 2SO2(g)+O2(g)
Calculate Keq for this reaction if the equalibrium concentrations are: |SO2|=0.42M; |O2|=0.21M; |SO3|=0.072M

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 23.06.2019 14:30, sha273
Will give imagine you are given a mystery element. it is, however, a discovered and known element. you may perform a maximum of two observations or tests to determine its identity. time and money is critical, so you need to prioritize your tests. if you can get by with a single test, you get 100 super-geek points from your research lab team. pick your two tests, number them as #1 and #2, and justify why you think these two will certainly be enough (and why the first might well be enough all by itself.) the available tests are classification into metal, non-metal, or metalloid, count of valence electrons, count of electron shells, atomic radius (error range: +/- 1 pm), electronegativity (error range: +/- 0.1), first ionization energy (error range: +/- 10 kj/mole), melting point (error range: +/- 10 c), and boiling point (error range: +/- 20 c).
Answers: 2
You know the right answer?
Questions in other subjects:










![\left [ SO_{2} \right ] = 0.42 \textrm{ M}, \left [ O_{2} \right ] = 0.21 \textrm{ M}, \left [ SO_{3} \right ] = 0.072 \textrm{ M}](/tpl/images/0549/0905/fc0fc.png) \\
\\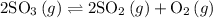
![K_{eq} = \displaystyle \frac{\left [ SO_{2} \right ]^{2}\times \left [ O_{2} \right ]}{\left [ SO_{3} \right ]^{2}} \\K_{eq} = \displaystyle \frac{\left ( 0.42 \right )^{2}\times 0.21}{\left ( 0.072 \right )^{2}} \\K_{eq} = 7.145](/tpl/images/0549/0905/2d355.png)
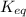 for the reaction is 7.145
for the reaction is 7.145 


