
Chemistry, 16.03.2020 20:27 ronniethefun
In a coffee-cup calorimeter experiment, 10.00 g of a soluble ionic compound was added to the calorimeter containing 75.0 g H2O initially at 23.2°C. The temperature of the water increased to 31.8°C. What was the change in enthalpy for the dissolution of the compound? Give your answer in units of joules per gram of compound. Assume that the specific heat of the solution is the same as that of pure water, 4.18 J ⁄ (g ⋅ °C).

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, haileywebb8
If you want to create an electrical current, which situation would produce a solution capable of this
Answers: 3

Chemistry, 22.06.2019 06:00, rebeccacruzz2017
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

You know the right answer?
In a coffee-cup calorimeter experiment, 10.00 g of a soluble ionic compound was added to the calorim...
Questions in other subjects:

Mathematics, 19.07.2019 20:00



Biology, 19.07.2019 20:00

Mathematics, 19.07.2019 20:00



Biology, 19.07.2019 20:00

Social Studies, 19.07.2019 20:00

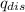 ) is equal to the heat of water (
) is equal to the heat of water (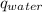 ).
). 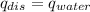 = ΔH/m= 2696.1 J/10.00 g= 269.61 J/g
= ΔH/m= 2696.1 J/10.00 g= 269.61 J/g

