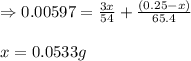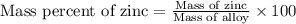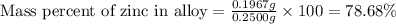Chemistry, 16.03.2020 19:21 juliannabartra
A 0.2500 g sample of an alloy reacts with to form hydrogen gas: 2Al(s) + 6H+(aq) 2Al3+(aq) + 3H2(g) Zn(s) + 2H+(aq) Zn2+(aq) + H2(g) The hydrogen produced has a volume of 0.147 L at 25ºC and 755 mm Hg. What is the percentage of zinc in the alloy

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:50, kyleighmarie05
H2so4(aq) + mg(s)—> mgso4(aq) +h2(g) which substance is the acid in the reaction?
Answers: 3

Chemistry, 22.06.2019 05:30, nuclearfire278
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease. correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 23:20, svaskeacevilles5477
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2

Chemistry, 23.06.2019 00:30, tateandvioletAHS14AY
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
You know the right answer?
A 0.2500 g sample of an alloy reacts with to form hydrogen gas: 2Al(s) + 6H+(aq) 2Al3+(aq) + 3H2(g)...
Questions in other subjects:




Physics, 17.10.2019 12:00

History, 17.10.2019 12:00


Mathematics, 17.10.2019 12:00



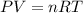
![25^oC=[25+273]K=298K](/tpl/images/0548/8750/df1f6.png)
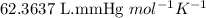

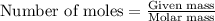 .....(1)
.....(1)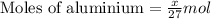
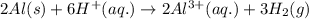
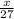 moles of aluminium will produce =
moles of aluminium will produce = 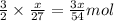 of hydrogen gas
of hydrogen gas
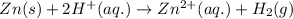
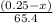 moles of zinc will produce =
moles of zinc will produce = 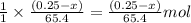 of hydrogen gas
of hydrogen gas