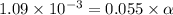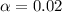Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, lindseyklewis1p56uvi
A+b→2c when the reaction begins, the researcher records that the rate of reaction is such that 1 mole of a is consumed per minute. after making changes to the reaction, the researcher notes that 2 moles of a are consumed per minute. what change could the researcher have made to effect this change?
Answers: 1

Chemistry, 22.06.2019 04:30, only1cache
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3

Chemistry, 22.06.2019 05:30, medlinalex
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
You know the right answer?
) PABA refers to para-aminobenzoic acid which is used in some sunscreen formulations. If a 0.055 M s...
Questions in other subjects:


Mathematics, 27.08.2019 21:10

Health, 27.08.2019 21:10

Chemistry, 27.08.2019 21:20

Chemistry, 27.08.2019 21:20

Business, 27.08.2019 21:20

Mathematics, 27.08.2019 21:20


Health, 27.08.2019 21:20

Mathematics, 27.08.2019 21:20

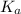 of PABA is 0.000022
of PABA is 0.000022



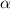 = ?
= ?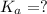
![pH=-log[H^+]](/tpl/images/0548/7597/15713.png)
![2.96=-log[H^+]](/tpl/images/0548/7597/5022f.png)
![[H^+]=1.09\times 10^{-3}](/tpl/images/0548/7597/3aec8.png)
![[H^+]=c\times \alpha](/tpl/images/0548/7597/4fc41.png)