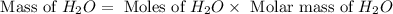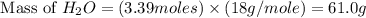Chemistry, 16.03.2020 17:58 BARRION1981
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many modern chemical processes. In the first step of the Ostwald process, ammonia is reacted with oxygen gas to produce nitric oxide and water. What is the maximum mass of H 2 O that can be produced by combining 90.6 g of each reactant? 4 NH 3 ( g ) + 5 O 2 ( g ) ⟶ 4 NO ( g ) + 6 H 2 O ( g )

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:40, jude3412
In an effort to address concerns about global warming, a power plant in portland, oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1


Chemistry, 22.06.2019 12:00, vannitling12p4w44f
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1
You know the right answer?
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many mod...
Questions in other subjects:

Mathematics, 25.09.2019 20:30

Geography, 25.09.2019 20:30



Physics, 25.09.2019 20:30

Chemistry, 25.09.2019 20:30



Mathematics, 25.09.2019 20:30

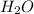 produced is, 61.0 grams.
produced is, 61.0 grams.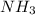 = 90.6 g
= 90.6 g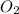 = 90.6 g
= 90.6 g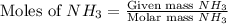
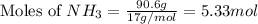
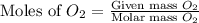
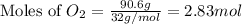
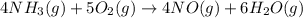
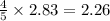 moles of
moles of 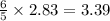 mole of
mole of