
You are given 10.00 mL of a solution of an unknown acid. The pH of this solution is exactly 2.95. You determine that the concentration of the unknown acid was 0.1224 M. You also determined that the acid was monoprotic (HA). What is the K_a and pK_a of your unknown acid

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 07:00, shradhwaip2426
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 08:00, hdjsjfjruejchhehd
Define dew point. i am writing this part to be able to ask the question
Answers: 1
You know the right answer?
You are given 10.00 mL of a solution of an unknown acid. The pH of this solution is exactly 2.95. Yo...
Questions in other subjects:










 is dissociation constant and the value of
is dissociation constant and the value of 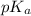 is 4.98.
is 4.98.![pH=-\log[H^+]](/tpl/images/0548/6523/cf945.png)
![2.95=-\log[H^+]](/tpl/images/0548/6523/b4bb5.png)
![[H^+]=10^{-2.95}=0.001122 M](/tpl/images/0548/6523/2ad17.png) ..[1]
..[1]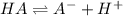
![K_a=\frac{[A^-][H^+]}{[HA]}](/tpl/images/0548/6523/a5cb9.png)
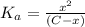
![[H^+]=x =0.001122 M](/tpl/images/0548/6523/90707.png) ( from [1])
( from [1])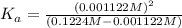
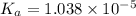
![pK_a=-\log[K_a]](/tpl/images/0548/6523/78bbf.png)
![=-\log[1.038\times 10^{-5}]=4.98](/tpl/images/0548/6523/44380.png)



