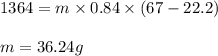Chemistry, 13.03.2020 02:28 alannaamarriee
A sample of sand initially at 22.2 °C absorbs 1364 J of heat. The final temperature of the sand is 67 °C. What is the mass (in g) of sand in the sample? The heat capacity of sand is 0.84

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:00, sandeebassett3
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 19:50, strawberrymrmr756
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3
You know the right answer?
A sample of sand initially at 22.2 °C absorbs 1364 J of heat. The final temperature of the sand is 6...
Questions in other subjects:

English, 16.09.2021 04:10


English, 16.09.2021 04:10

Mathematics, 16.09.2021 04:10


Mathematics, 16.09.2021 04:10



History, 16.09.2021 04:10

Mathematics, 16.09.2021 04:10

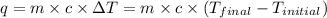
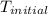 = initial temperature = 22.2°C
= initial temperature = 22.2°C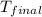 = final temperature = 67°C
= final temperature = 67°C