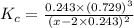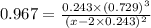The equilibrium constant, Kc, for the following reaction is 0.967 at 650 K. 2NH3(g) N2(g) 3H2(g) When a sufficiently large sample of NH3(g) is introduced into an evacuated vessel at 650 K, the equilibrium concentration of H2(g) is found to be 0.729 M. Calculate the concentration of NH3 in the equilibrium mixture. M

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 22:30, robertss403
How many moles of kci are produced from 2.50 moles k
Answers: 1
You know the right answer?
The equilibrium constant, Kc, for the following reaction is 0.967 at 650 K. 2NH3(g) N2(g) 3H2(g) Whe...
Questions in other subjects:

Spanish, 01.10.2019 06:30


English, 01.10.2019 06:30

Health, 01.10.2019 06:30


Mathematics, 01.10.2019 06:30

Biology, 01.10.2019 06:30



 in the equilibrium mixture is 0.31 M
in the equilibrium mixture is 0.31 M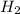 = 0.729 M
= 0.729 M
![K_c=\frac{[y]\times [3y]^3}{[x-2y]^2}](/tpl/images/0544/6710/3f7db.png)