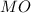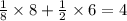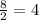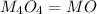Chemistry, 12.03.2020 01:46 Tcareyoliver
A solid metal oxide crystallizes in a cubic unit cell. In the unit cell there are metal (M) ions on every corner and in the center of every face. Oxide ions (O) are found in half of the tetrahedral holes. What is the empirical formula of this metal oxide?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, cicimarie2018
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1


Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 23:20, svaskeacevilles5477
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2
You know the right answer?
A solid metal oxide crystallizes in a cubic unit cell. In the unit cell there are metal (M) ions on...
Questions in other subjects:

Mathematics, 23.06.2019 22:00




Mathematics, 23.06.2019 22:00

History, 23.06.2019 22:00



Physics, 23.06.2019 22:00