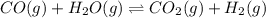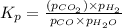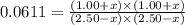Chemistry, 12.03.2020 00:02 Mitchmorgan3816
CO(g ) + H2O(g ) <---> CO2(g ) + H2(g ), Kc = 0.0611 at 2000 K . A reaction mixture initially contains a CO partial pressure of 2.50 atm, an H2O partial pressure of 2.50 atm, a CO2 partial pressure of 1.00 atm, and an H2 partial pressure of 1.00 atm at 2000 K. Calculate the equilibrium partial pressure of CO

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, ayoismeisalex
Which of these sequences lists the correct order for the creation of sedimentary rock from sediment? a. deposition, burial, compaction, cementation b. burial, deposition, compaction, cementation c. compaction, deposition, burial, cementation d. cementation, deposition, burial, compaction
Answers: 1


Chemistry, 22.06.2019 17:30, kevin72937
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1

Chemistry, 22.06.2019 21:00, estherdinhllama
Read "who built the pyramids? ”. leave this link open while you answer the questions throughout the assignment. give at least two reasons why some people claim the pyramids of giza were constructed by aliens.
Answers: 1
You know the right answer?
CO(g ) + H2O(g ) <---> CO2(g ) + H2(g ), Kc = 0.0611 at 2000 K . A reaction mixture initially...
Questions in other subjects:


Mathematics, 08.12.2020 20:20


Mathematics, 08.12.2020 20:20




Chemistry, 08.12.2020 20:20

Mathematics, 08.12.2020 20:20

 is the constant of a certain reaction at equilibrium.
is the constant of a certain reaction at equilibrium.