
Chemistry, 11.03.2020 22:56 amberpetty4288
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that goes with this equilibrium constant. (Use H3O+ instead of H+.)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, ulilliareinhart2
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 04:30, mamabates181981
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 09:00, heids17043
Chen drew a diagram to compare the ways in which different organisms obtain nitrogen. which label belongs to the area marked z?
Answers: 3
You know the right answer?
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that...
Questions in other subjects:


Mathematics, 06.09.2019 11:10

Spanish, 06.09.2019 11:10

History, 06.09.2019 11:10

History, 06.09.2019 11:10

Computers and Technology, 06.09.2019 11:10

Mathematics, 06.09.2019 11:10



History, 06.09.2019 11:10

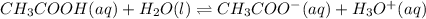
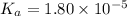
![K_c=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH][H_2O]}](/tpl/images/0543/5879/7c275.png)
![K_a=K_c\times [H_2O]=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/125a5.png)
![[H_2O]=1](/tpl/images/0543/5879/b8579.png)
![K_a=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/e4df0.png)


