
Chemistry, 11.03.2020 00:24 MarishaTucker
Carbon monoxide gas reacts with hydrogen gas to form methanol via the following reaction: CO(g)+2H2(g)→CH3OH(g)CO(g)+2H2(g)→C H3OH(g) A 1.55 LL reaction vessel, initially at 305 KK, contains carbon monoxide gas at a partial pressure of 232 mmHgmmHg and hydrogen gas at a partial pressure of 389 mmHgmmHg . Identify the limiting reactant and determine the theoretical yeild of methonal in grams.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, cheesecake1919
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 16:00, bbrogle5154
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1

Chemistry, 22.06.2019 16:50, brandiwingard
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2
You know the right answer?
Carbon monoxide gas reacts with hydrogen gas to form methanol via the following reaction: CO(g)+2H2(...
Questions in other subjects:


Social Studies, 01.07.2019 06:20


Biology, 01.07.2019 06:20

Biology, 01.07.2019 06:20

Social Studies, 01.07.2019 06:20


Mathematics, 01.07.2019 06:20

Mathematics, 01.07.2019 06:20

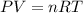 ..........(1)
..........(1)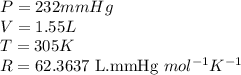
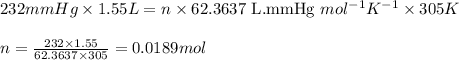
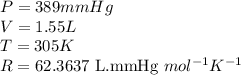
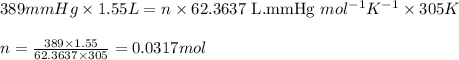
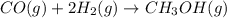
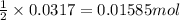 of carbon monoxide
of carbon monoxide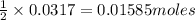 of methanol
of methanol



