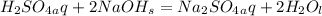Problem PageQuestion Aqueous sulfuric acid will react with solid sodium hydroxide to produce aqueous sodium sulfate and liquid water . Suppose 91. g of sulfuric acid is mixed with 116. g of sodium hydroxide. Calculate the maximum mass of sodium sulfate that could be produced by the chemical reaction. Round your answer to significant digits.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, jzjajsbdb8035
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 23.06.2019 00:50, lakhanir2013
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3

Chemistry, 23.06.2019 04:31, laurenbreellamerritt
How big are the bighest ocean waves at mavericks
Answers: 1
You know the right answer?
Problem PageQuestion Aqueous sulfuric acid will react with solid sodium hydroxide to produce aqueous...
Questions in other subjects:





Physics, 17.05.2020 13:57

Mathematics, 17.05.2020 13:57

Social Studies, 17.05.2020 13:57



Mathematics, 17.05.2020 13:57