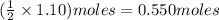Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, tdowling331
What must happen before a body cell can begin mitotic cell division
Answers: 2

Chemistry, 22.06.2019 13:30, kassandrarosario1115
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1

Chemistry, 22.06.2019 15:30, ashtonviceoxd21i
The gulf stream is a warm water current that flows away from the equator to northern europe. witch of these does it cause. a. crashes of warm and cool water in the ocean b. colder climates near the equator c. large waves on the cost of europe d. warm climates in northern europe
Answers: 1

Chemistry, 22.06.2019 20:10, maddie1776
Insoluble sulfide compounds are generally black in color. which of the following combinations could yield a black precipitate? check all that apply. na2s(aq)+kcl(aq) li2s(aq)+pb(no3)2(aq) pb(clo3)2(aq)+nano3(aq) agno3(aq)+kcl(aq) k2s(aq)+sn(no3)4(aq)
Answers: 1
You know the right answer?
Consider the balanced equation. 2HCl + Mg MgCl2 + H2 If 40.0 g of HCl react with an excess of magnes...
Questions in other subjects:



Geography, 09.12.2020 18:10

Physics, 09.12.2020 18:10

English, 09.12.2020 18:10

Physics, 09.12.2020 18:10


History, 09.12.2020 18:10

English, 09.12.2020 18:10

Mathematics, 09.12.2020 18:10

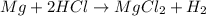
 .
.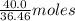 of HCl = 1.10 moles of HCl
of HCl = 1.10 moles of HCl