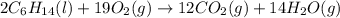Chemistry, 10.03.2020 04:26 spazzinchicago
List the number of each type of atom on the left side of the equation 2C6H14(l)+19O2(g)→12CO2(g)+14H2O(g) Enter your answers separated by commas (the order of the numbers is the same as the order of the elements on the left side of the equation).

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:00, PrincessKeliah5538
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 10:00, emfranco1
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2
You know the right answer?
List the number of each type of atom on the left side of the equation 2C6H14(l)+19O2(g)→12CO2(g)+14H...
Questions in other subjects:





English, 13.06.2020 23:57

English, 13.06.2020 23:57

Mathematics, 13.06.2020 23:57

Mathematics, 13.06.2020 23:57

History, 13.06.2020 23:57

Spanish, 13.06.2020 23:57