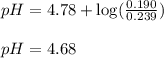Chemistry, 10.03.2020 00:07 laurabwhiddon
Calculate the pH of a solution that is 0.239 M acetic acid and 0.190 M sodium acetate. The Ka of acetic acid is 1.76×10–5 at 25°C. What is the pH of this mixture at 0°C? (At 0°C, Ka = 1.64x10-5)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:10, leo4687
What does a particular point on a line of a phase diagram represent? o a. the maximum temperature a substance can exist at without bonds breaking b. the pressure created by the kinetic energy of molecules at a particular temperature c. the melting point or boiling point of a substance at a specific pressure d. the conditions in which temperature and pressure have equal effects on a substance
Answers: 2

Chemistry, 22.06.2019 04:30, earcake2470
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1


Chemistry, 22.06.2019 08:00, flakko1899
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1
You know the right answer?
Calculate the pH of a solution that is 0.239 M acetic acid and 0.190 M sodium acetate. The Ka of ace...
Questions in other subjects:



Mathematics, 05.11.2021 19:40






History, 05.11.2021 19:40

![pH=pK_a+\log(\frac{[salt]}{[acid]})](/tpl/images/0539/1827/e4eea.png)
![pH=pK_a+\log(\frac{[CH_3COONa]}{[CH_3COOH]})](/tpl/images/0539/1827/05ea7.png)
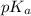 = negative logarithm of acid dissociation constant of acetic acid at 0°C = 4.78
= negative logarithm of acid dissociation constant of acetic acid at 0°C = 4.78![[CH_3COONa]=0.190M](/tpl/images/0539/1827/29d58.png)
![[CH_3COOH]=0.239M](/tpl/images/0539/1827/577dd.png)