
Chemistry, 09.03.2020 23:58 denaeyafranklin8430
Calculate the enthalpy of formation (kJ/mol) of MgO(s). The enthalpy of reaction for the equation as written is 100.8 kJ/mol. If the answer is negative, enter the sign and then the magnitude.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kiki197701
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3


Chemistry, 22.06.2019 15:30, neariah24
Plz me ! 1 which of earths spheres contains most of its mass? a atmosphere b hydrosphere c geosphere* d biosphere 2 erosion and weathering are examples of which types of forces? a constructive forces b destructive forces* c gravitational forces d inertia-related forces 3 which of the following statements about earths atmosphere is true? a earths atmosphere contains 78% water vapor which is essentail to life b earths atmosphere contains 21% oxygen c earths atmosphere contains carbon dioxide which all life forms require d earths atmosphere allows radiation from the sun to pass through it and warm earths surface* 4 the strenght of the force of gravity between two objects is determined by which of the following factors? select all that apply a the messes of the objects* b the distance between the objects* c the volumes of the objects d the surface area of the objects 5 earth and moon are kept in there respective orbits due to the influence of a inertia b gravity c gravity and inertia* d neither gravity or inertia if you answer all questions right i will give
Answers: 1

Chemistry, 23.06.2019 01:00, williedenmark42
What is the most common form of matter in the universe
Answers: 2
You know the right answer?
Calculate the enthalpy of formation (kJ/mol) of MgO(s). The enthalpy of reaction for the equation as...
Questions in other subjects:


Mathematics, 21.10.2020 01:01

English, 21.10.2020 01:01

Mathematics, 21.10.2020 01:01



Mathematics, 21.10.2020 01:01



Mathematics, 21.10.2020 01:01

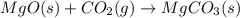
![\Delta H^o_{rxn}=\sum [n\times \Delta H_f_{(product)}]-\sum [n\times \Delta H_f_{(reactant)}]](/tpl/images/0539/1394/e893d.png)
![\Delta H_{rxn}=[(1\times \Delta H_f_{(MgCO_3(s))})]-[(1\times \Delta H_f_{(MgO(s))})+(1\times \Delta H_f_{(CO_2(g))})]](/tpl/images/0539/1394/01988.png)
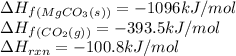
![-100.8=[(1\times (-1096))]-[(1\times \Delta H_f_{(MgO(s))})+(1\times (-393.5))]\\\\\Delta H_f_{(MgO(s))}=-601.7kJ/mol](/tpl/images/0539/1394/d8103.png)


