
Chemistry, 07.03.2020 07:31 GabbyLandry
The molar mass of a compound is 92 g/mol. Analysis of a sample of the compound indicates that it contains 0.606g of N and 1.390g of O. Find its molecular formula.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3


Chemistry, 23.06.2019 01:50, UncleVictor5188
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1
You know the right answer?
The molar mass of a compound is 92 g/mol. Analysis of a sample of the compound indicates that it con...
Questions in other subjects:

History, 16.08.2021 22:40



Mathematics, 16.08.2021 22:40



Spanish, 16.08.2021 22:40


Mathematics, 16.08.2021 22:40


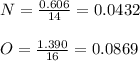
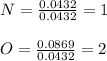
![[NO2]n = 92[14 + (16x2)]n = 92[14 +32]n = 9246n = 92](/tpl/images/0538/1065/5116b.png)
![[NO2]n \\= [NO2]2\\ = N2O4](/tpl/images/0538/1065/6f423.png)


