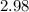Chemistry, 07.03.2020 06:17 missheather0309
A sample of solid NH4NO3 was placed in an evacuated container and then heated so that it decomposed explosively according to the following equation:
NH4NO3(s) N2O(g) + 2H2O(g)
At equilibrium, the total pressure in the container was found to be 2.72 bar at a temperature of 500.°C. Calculate Kp.
a.
1.64
b.
0.822
c.
2.98
d.
80.5
e.
0.745

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, yousifgorgees101
The earth's moon is unusually large. two popular theories of the moon's origin include the "sister world" hypothesis, which states that the moon formed from the same materials as the earth, near enough to the earth that they fell into orbit around each other. a second theory is the "capture" hypothesis, in which the moon formed elsewhere in the solar system, and the earth's gravity pulled it into its orbit. studies of what the moon is made of indicate that some of its materials had to come from the earth or from the same area of the solar system where the earth had formed. at the same time, the moon does not contain much of the material that makes up the earth's core, so the moon could not have formed from the same materials as the earth. how do the two facts above affect the described theories of the moon's origin? a. they show that scientists will never agree on where the moon came from. b. they show that more experiments on moon formation need to be done. c. they show that no theory accounts for the existence of the moon. d. they show that neither theory is complete and entirely correct.
Answers: 1

Chemistry, 22.06.2019 19:30, gracieisweird12
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 23.06.2019 04:00, anonymous1813
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2

You know the right answer?
A sample of solid NH4NO3 was placed in an evacuated container and then heated so that it decomposed...
Questions in other subjects:

History, 10.12.2020 14:00









Mathematics, 10.12.2020 14:00