
Chemistry, 07.03.2020 05:21 juanitarodriguez
Calculate the entropy change when a. two moles of H2O(g) are cooled irreversible at constant p from 120°C to 100°C. b. one mole of H2O(g) is expanded at constant pressure of 2 bar from an original volume of 20 L to a final volume of 25 L. You can consider the gas to be ideal. c. one hundred grams of H2O(s) at -10°C and 1 bar are heated to H2O(l) at +10°C and 1 bar.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, tcapele252
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 21:00, lalaween098
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

You know the right answer?
Calculate the entropy change when a. two moles of H2O(g) are cooled irreversible at constant p from...
Questions in other subjects:





Mathematics, 26.08.2020 01:01

History, 26.08.2020 01:01

History, 26.08.2020 01:01

Mathematics, 26.08.2020 01:01

Mathematics, 26.08.2020 01:01

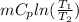 Where
Where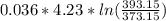 = 0.00795 kJ/(K)
= 0.00795 kJ/(K) 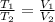 Therefore change in entropy S₂ - S₁ =
Therefore change in entropy S₂ - S₁ =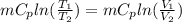
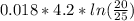 = -0.01686965 kJ/(K) = -16.9 J/K
= -0.01686965 kJ/(K) = -16.9 J/K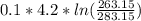 = -0.03078 kJ/K
= -0.03078 kJ/K

