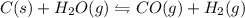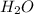Chemistry, 07.03.2020 04:48 katherine3084
Coal can be used to generate hydrogen gas (a potential fuel) by thefollowing endothermic reaction. C(s) + H2O (g) <==> CO(g) + H2(g)if this reaction mixture is at equilibrium, predict whether each ofthe following will result in the formation of additional hydrogengas, the formation of less hydrogen gas, or have no effect on thequantity og hydrogen gas. a. adding more C to the reaction mixtureb. adding more H2O to the reaction mixturec. raising the temperature of the reaction mixtured. increasing the volume of the reaction mixturee. adding a catalyst to the reaction mixturef. adding an inert gas to the reaction mixture

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 23.06.2019 09:10, amandapill
In a 28 g serving of cheese curls there are 247mg of sodium. how much sodium is in a 12.5 ounce bag
Answers: 1
You know the right answer?
Coal can be used to generate hydrogen gas (a potential fuel) by thefollowing endothermic reaction. C...
Questions in other subjects:

Mathematics, 20.09.2020 17:01


Mathematics, 20.09.2020 17:01

Chemistry, 20.09.2020 17:01


Mathematics, 20.09.2020 17:01

Mathematics, 20.09.2020 17:01

Physics, 20.09.2020 17:01