
Chemistry, 07.03.2020 04:14 mckleinrivero
The rate constant for a certain reaction is kkk = 3.50×10−3 s−1s−1 . If the initial reactant concentration was 0.450 MM, what will the concentration be after 19.0 minutes?
A zero-order reaction has a constant rate of 3.50×10−4 M/sM/s. If after 65.0 seconds the concentration has dropped to 3.50×10−2 MM, what was the initial concentration?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:00, kayla32213
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1

Chemistry, 23.06.2019 12:50, almaga1979orfvwo
Complete the paragraph to describe the characteristics of a borane molecule (bh3). the lewis structure and table of electronegativities are given. the bond polarities in bh3 are , the molecular shape is , and the molecule is .
Answers: 2

You know the right answer?
The rate constant for a certain reaction is kkk = 3.50×10−3 s−1s−1 . If the initial reactant concent...
Questions in other subjects:

Mathematics, 12.05.2021 20:30

Mathematics, 12.05.2021 20:30

Mathematics, 12.05.2021 20:30


Chemistry, 12.05.2021 20:30


Mathematics, 12.05.2021 20:30



![Rate = k[A][B]](/tpl/images/0537/5969/ab111.png) . As the data are given in the question, the rate constant is
. As the data are given in the question, the rate constant is 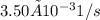 , the initial concentration
, the initial concentration![[A]_o = 0.450 M](/tpl/images/0537/5969/1d14c.png) and time is 19 minutes that is the 1140s.
and time is 19 minutes that is the 1140s.
![ln[A] = ln[A]_o - kt\\ln[A] = ln (0.450) - (3.50*10^-^3)1140\\ln[A] = -0.799 - 3.99\\ln[A] = -4.789\\[A] = 0.0083M](/tpl/images/0537/5969/91bfd.png)
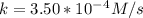 Time = 65 s
Final Concentration [A] =
Time = 65 s
Final Concentration [A] = 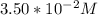
![[A] = [A]_o - kt[A]_o = [A] + kt\\[A]o = 3.50*10^{-2} + (3.50*10^{-4}) * 65\\[A]_o = 3.50*10^{-2} + 0.02275\\[A]_o = 0.05775 M](/tpl/images/0537/5969/3e5cb.png)



